39 warning labels on drugs
FDA Drug Safety Communication: FDA strengthens warning that ... The U.S. Food and Drug Administration (FDA) is strengthening an existing label warning that non-aspirin nonsteroidal anti-inflammatory drugs (NSAIDs) increase the chance of a heart attack or stroke. Pharmaceutical Labeling 101: FDA Regulations Guide Boxed Warning: The label should now include a boxed warning, stating possible adverse drug reactions and other health risks from taking the drug. Recent Major Changes: A new section called "Recent Major Changes" has been introduced. This section aims to inform the reader about the changes made in FPI.
FDALabel: Full-Text Search of Drug Product Labeling | FDA Labeling for approved OTC drugs and OTC drugs under the monograph system is called Drug Facts. Drug Facts includes information about the purpose and use of the drug, warnings, directions for use,...

Warning labels on drugs
Drug Warning Label Pictures, Images and Stock Photos drug warning label stock pictures, royalty-free photos & images. Prescription Drugs and Driving. A bottle of prescription drugs on a white background. The open pill bottle is spilling a variety of pills out onto the white surface with a set of car keys beside it. This concept illustrates driving under prescription drugs, abusing narcotics, DUI ... Perceptions of prescription warning labels within an underserved ... Prescription warning labels are small colored stickers placed adjacent to the drug label on a prescription bottle that provides important cautionary information concerning the safe administration of a medicine. For example, "take with food" or "limit time in sunlight when taking this medication". Adults Over 50 Often Ignore Prescription Drug Warning Labels The warning labels include instructions such as, "Do not drive while taking this medication," or "Avoid smoking while taking this drug," the Los Angeles Times reports. The study, published in the journal PLoS One, found participants over age 50 were much less likely to pay attention to the warning labels than those ages 20 to 29.
Warning labels on drugs. Prescription Drug Labels Don't Have To Confuse | Learn More Here The FDA reserves its most serious warning, the boxed warning, for drugs or medical devices carrying what it deems to be a significant risk of serious or life-threatening side effects. Such a warning doesn't necessarily render a drug unsafe, but it does warrant a conversation between patients and prescribing physicians. Black Box Warnings - Fast-Tracked Drugs & Increased Use A black box warning is the FDA's most stringent warning for drugs and medical devices on the market. Black box warnings, or boxed warnings, alert the public and health care providers to serious side effects, such as injury or death. The FDA requires drug companies to add a warning label to medications that have a black box warning. FDA Black Box Warnings of 8 Very Common Drugs: Read Your Labels Mar 23, 2017 · A black box warning is a consumer warnings with a black border placed on labels calling out the product’s serious health risks—like the one that appears on tobacco packaging. It is the most severe type of warning by the Food and Drug Administration (FDA). Cigarette Labeling and Health Warning Requirements | FDA Cigarette Packages. Size and location – The required warning must comprise at least the top 50 percent of the front and rear panels of the cigarette package (i.e., the two largest sides or ...
Black Box Drugs | FDA Warning Information Uloric: This gout drug created by Takeda Pharmaceuticals received a black box warning in February 2019, after studies revealed that it contributed to more cardiac-related incidents than its most common competitor, allopurinol. As a result, some people have started filing Uloric lawsuits. 438 Drug Warning Label Stock Photos - Dreamstime Close-up of a label on a bottle of prescription medication warning not to consume alcohol while using the drug Customer in pharmacy holding medicine bottle. Woman reading the label text about medical information or side effects in drug store. Stanton Introduces Legislation to Require Warning Labels on Addictive ... The bill would amend the Controlled Substance Act to require the Food and Drug Administration to issue regulations to require warning labels be added directly to the opioid prescription bottle that states that the drugs may cause "dependence, addiction, or overdose." This is similar to what is required for cigarette packaging. A Warning About Warning Labels - U.S. Pharmacist Label Color: Approximately 42% of the patients correlated the label's color to the severity of the message. Patients reported that a red label meant danger, yellow translated to caution, and blue, white, and green labels were viewed as "recommendations" that were not as critical as the instructions on red labels.
Drug Warning Labels: The Good, Bad, and Ugly | The Motley Fool The Food and Drug Administration can influence how well a drug sells by determining the warning labels that must accompany the drug. Unfortunately for drugmakers, the ability to slap a warning ... Who Reads the Drug Warning Labels?- Hormones Matter Perhaps doctors and patients alike are doing the sensible thing in not bothering to read drug warning labels. If the information that they give is arbitrary and they don't help people to assess the actual risk associated with a drug properly, they should be ignored. The only party that the warning labels are truly serving is the drug ... Understanding the FDA's Black Box Warnings on Drug Labels Boxed warnings, also known as black box warnings, are the most serious type of warning issued by the Food and Drug Administration (FDA). These warnings are front and center on a drug's package... What Is A FDA Black Box Warning Label - NastLaw Patients and doctors rely on proper warning labels on drugs. Black box warning labels are one way to keep the public safe, but they are often added long after the drug has been on the market and many people have suffered serious side effects. Drugs that caused serious side effects without providing proper warnings are often the subject of ...
The Importance of Reading Medication Warning Labels Another common warning label on prescriptions is "May cause photosensitivity; avoid sunlight while taking this medication". You may think you are safe because you work in an office and don't spend much time outside, but even the few minutes of direct sunlight you receive while driving can trigger a reaction.
Warning label - Wikipedia A warning label is a label attached to a product, or contained in a product's instruction manual, warning the user about risks associated with its use, and may include restrictions by the manufacturer or seller on certain uses. ... Drug, and Cosmetic Act of 1938.
325 DOT Hazardous Materials Warning Labels and Markings ... The warning labels shown in Exhibit 325.3 a, Exhibit 325.3 b, and Exhibit 325.4 may appear only on mailpieces containing mailable hazardous materials that require use of the label under Postal Service requirements. Division 5.1, 5.2, Class 8 and Class 9 labels are only permitted when used in conjunction with a Limited Quantity air mark.
Pharmacy, Medication Warning Labels & Stickers | PDC Never get important medications or directions mixed up ever again with these pre-printed warning labels for medical, pharmacy, and lab applications. ... Communication Label (Paper, Permanent) Drug Discontinued 1-9/16" x 3/8" Yellow - 500 per Roll, 2 Rolls per Box . 1-378. $7.05. Add to Cart.
Boxed warning - Wikipedia The FDA can require a pharmaceutical company to place a boxed warning on the labeling of a prescription drug, or in literature describing it. It is the strongest warning that the FDA requires, and signifies that medical studies indicate that the drug carries a significant risk of serious or even life-threatening adverse effects.
Warning Label Templates - Customizable Templates | Avery.com Select the candle warning label template below to fit your candle containers. Our round labels are completely editable. Add your logo to small rectangles or choose a tag warning label to hang around your candle. 1" x 2-5/8" Rectangle Label 2-1/2" Round Label 2-1/2" Round Label 2" x 3" Rectangle Label 1" x 2-5/8" Rectangle Label 2-1/2" Round Label
Defective Drug Warning Labels and Off-Label Use - LawInfo An adequate warning label for a drug will include information such as dosage, active ingredients, and known harmful side effects. However, it's often the case a drug is prescribed by a doctor for a use that wasn't intended, or hasn't been publically endorsed by the drug developers. This was the case with Fen-Phen. What about off-label use?
Do Drug Labels Overdo Warnings? - WebMD "These are 20,000- to 30,000-word massive documents." Warnings on Drug Labels In 2006, the FDA set rules to make drug labels easier to read and understand and reduce "overwarning" on drug labels,...
Prescription Labels and Drug Safety - Consumer Reports Warnings typed directly onto patient labels in a large typeface. Research has found that fewer than 10 percent of people examine their drug containers for the colorful warning stickers that...
Consumer Warning Labels Aren't Working Consumer Warning Labels Aren't Working by Lisa A. Robinson, W. Kip Viscusi, and Richard Zeckhauser November 30, 2016 Warning labels are everywhere. They alert us to the risks of eating unhealthy...
Misunderstanding Drug Warning Labels - Medscape These adhesive labels are widely used and provide important information regarding the safe administration of prescription medications. Failure to heed the warnings or special instructions on these...
50 Common Warning Labels On Medication Containers Top 50 Common Warning Labels and Their Meanings The medication must be swallowed whole. Because certain drugs are designed to be either fast-acting or slow-releasing, damaging the outer coating may lead to harmful damages to the body. The medication is intended for external use only. Ingesting it may lead to undesirable effects or even poisoning.
Labeling Information | Drug Products | FDA For prescription drug labeling resources (e.g., Prescribing Information, FDA-approved patient labeling, and carton and container labeling), please see the Prescription Drug Labeling Resources web page
Deceptive Product Label Case Found to Fall Under FDA Jurisdiction FDA gave warning letters to companies arguing methylsynephrine used in dietary supplements does not meet Federal Food, Drug, and Cosmetic Act FDCA dietary ingredient definition, making products ...
Adults Over 50 Often Ignore Prescription Drug Warning Labels - Partnership for Drug-Free Kids ...
Academic Journals | American Marketing Association Journal of Interactive Marketing aims to identify issues and frame ideas associated with the rapidly expanding field of interactive marketing, which includes both online and offline topics related to the analysis, targeting, and service of individual customers.
Flying Under the Influence: Commercial Pilots and Mind-altering Drugs—Playing Russian Roulette ...
Cautionary and advisory labels | About | BNF | NICE To be used on preparations containing ofloxacin and some other quinolones, doxycycline, lymecycline, minocycline, and penicillamine. These drugs chelate calcium, iron, and zinc and are less well absorbed when taken with calcium-containing antacids or preparations containing iron or zinc.

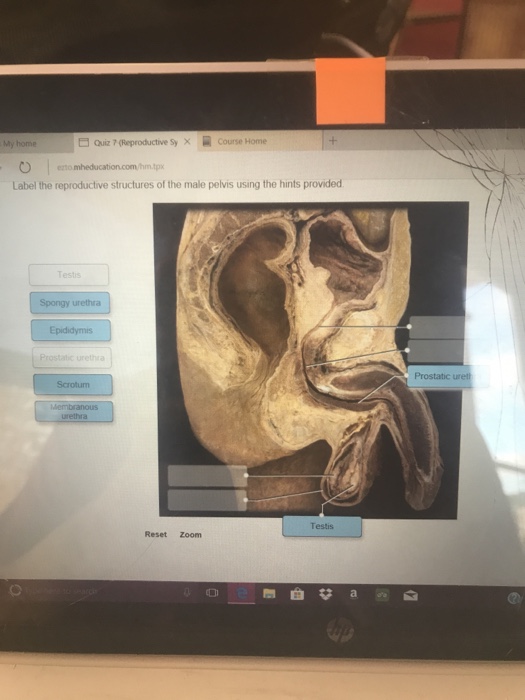








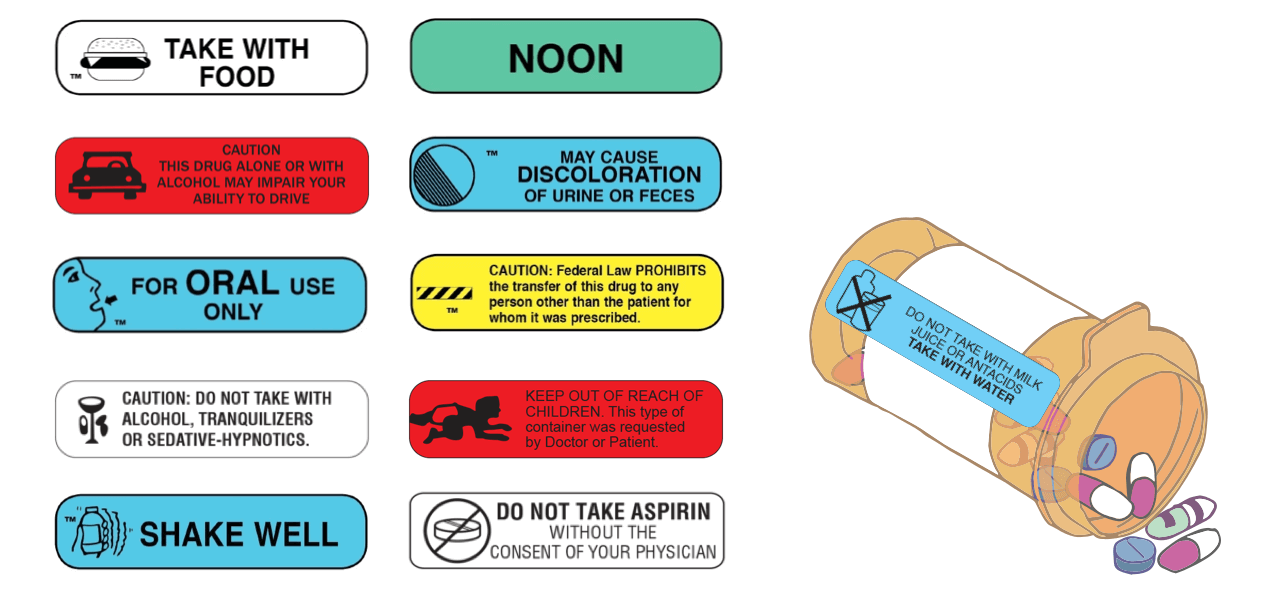
Post a Comment for "39 warning labels on drugs"